Body pH, training, and health
|
|
JNE wrote: One thing that can significantly effect the acidity inside your muscle tissue (or in any solution) is hydrogen. The more hydrogen the lower the pH (more acidic). During exercise, hydrogen is released in abundance as a result of breaking down fuels. Experiments have shown that the largest decreases in pH (muscles becoming more acidic) are during short bouts of intense, maximal effort that last anywhere from 1-10 minutes. That is one of the most poorly written pieces of pseudo-scientific nonsense I've read in a long time. Even if you look at it very ironically which you seem quite keen on doing. |
|
|
Boissal . wrote: Not to mention the accumulating hydrogen should provide some lift, albeit with the risk of exploding. Oh, the humanity! |
|
|
|
|
|
Boissal . wrote: Hydrogen ions are a byproduct of the ATP breakdown cycle during exercise (and yes, different than hydrogen ) . It can ramp up to very high levels of H+ during maximal effort. People can actually train in ways to improve their blood buffering abilities. Still, this has nothing to do with OP's original premise of diet....... I can haz energy? |
|
|
David K wrote:So? That doesn't prove anything at all about what you should eat. In fact, it's implied by that paragraph that the pH returns to normal after the short bout of maximal effort. No Way! Do you mean to suggest that the body can take nutrients from one part to use in another part! WOW! You must be a genius! Aerili wrote:Hard breathing is the body's response via the pulmonary system to rid itself of extra H+ ions. Which are typically formed from anaerobic exercise... which is usually considered "hard effort". Yes, and a hydrogen rich environment has what pH? And flooding a hydrogen-rich bloodstream with oxygen will do what again? |
|
|
Aerili wrote: So, if the body does not produce alkalinity (unlike acidity) naturally, then where does it get the stuff needed to buffer? Does it magically teleport itself? |
|
|
Oh, this thread is precious. |
|
|
JNE wrote: I am curious if anyone here pays attention to and is aware of the importance of the pH of their incoming nutrient stream. Specifically, the importance of eating plenty of alkaline foods, or foods which will digest into an alkaline state. Anyone notice any different training outcomes from balancing the pH of their diet? Any difference in health? Lol. Junk Science. Eat/drink natural food. Duh. |
|
|
Tradiban wrote: Nah, the 200 IQ move is to eat that delicious acidic diet and drink some bleach at the end of each day to compensate. Average pH is 9.4, baby! Can't argue with science. |
|
|
Ted Pinson wrote: Oh, this thread is precious. I made a mistake of reading this while eating. This thread made me hyperventilate, and when I laughed really hard, some honey lemon tea came up my nose. It hurts now — a clear proof that acidic foods will do you no good. |
|
|
|
|
|
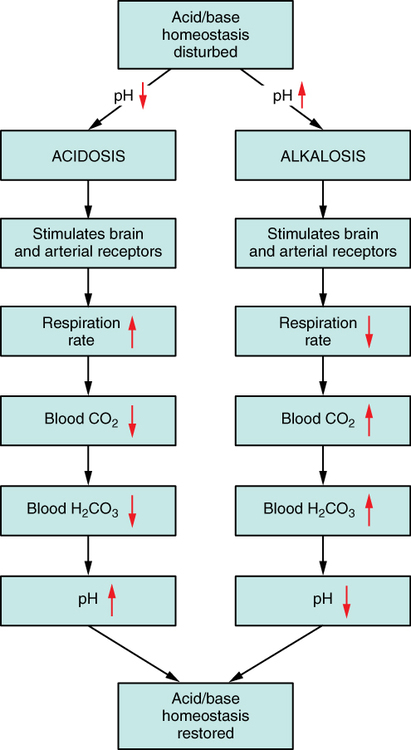
JNE wrote: This image conveniently answered your question before you asked it, when it was posted before: You may want to pay special attention to the left side of the diagram, which is where your question is answered. |
|
|
Because googling is too hard... |
|
|
All your base are belong to us. |
|
|
chris tregge wrote: I work in sales. Cool link about the specifics of how the kidneys regulate the bloods pH. Am I correct in reading that a "typical western diet" will lead to an overall acid load on the body, and that one of the mechanisms for dealing with that is for our body to produce parathyroid hormone in order to dump phosphate to be used as a urinary buffer for any titratable acid the kidneys need to get rid of? |
|
|
The OP should read and reflect upon this so I can get my endorsement check from Big Brother and Big Pharma $$$$$$$!!!!!!!!!! |
|
|
JNE wrote: David K answered your first question. It's called the acid-base transport system. As for question two, increased respiratory rate means you breath out the problematic ions. Respiration can adjust pH in minutes - much faster than the renal system. High respiration means you breathe out a lot of CO2, which rids the blood stream of carbonic acid, which means your pH gets higher, balancing the acidosis. HCO3- + H+ --> H2CO3 --> [ H2O + CO2(g) in the lungs] --> the CO2(g) is exhaled. Your hydrogen ions are now left behind forming water in the blood, reducing acidity. |
|
|
Aerili wrote:David K answered your first question. It's called the acid-base transport system. Actually, I give Ted Pinson credit for that one. And it was not a question on my part, it was an opportunity for the offending poster to demonstrate they were not completely ignorant regarding the subject at hand. Aerili wrote: As for question two, increased respiratory rate means you breath out the problematic ions. Respiration can adjust pH in minutes - much faster than the renal system. High respiration means you breathe out a lot of CO2, which rids the blood stream of carbonic acid, which means your pH gets higher, balancing the acidosis. HCO3- + H+ --> H2CO3 --> [ H2O + CO2(g) in the lungs] --> the CO2(g) is exhaled. Your hydrogen ions are now left behind forming water in the blood, reducing acidity. Your response appears to leave out the well established fact that a "typical western diet" will lead to an overall acidic load on all these buffering systems, and that in eating this "typical western diet" eventually these systems will wear out, leading to health problems, and that the fix to this (which is plainly obvious to any thinking thing) is in ones diet. |
|
|
Buffers don’t “wear out”. Physiology is complex, and misunderstood enough that silly unsubstantiated fads like the alkaline diet manage to take hold. |
|
|
Keenan Waeschle wrote: Buffers don’t “wear out”. So the body has an endless supply of internally produced phosphates to be used as a urinary buffer, and the things needed to produce this phosphate can be entirely gotten from a "typical western diet". Tell me more :) |

 Continue with onX Maps
Continue with onX Maps Sign in with Facebook
Sign in with Facebook