Bacterial corrosion of 304 (A2) rock anchors
|
|
David Reevewrote:From Wikipedia - Cayman Trough Wow, ya learn something everyday. Okay, so there's a good source for sulphates in the immediate area. But there's a possible mitigating factor, too. The Gulf Stream is ripping past the island at about 6 knots at the surface (right to left in the photo). Depending on where the vents are located, upstream or downstream, the sulphate might be low due to the exhaust from the vents being diluted. I suppose a sample is required. Just BTW: 450C water that's not steam because of the pressure is mind-boggling. |
|
|
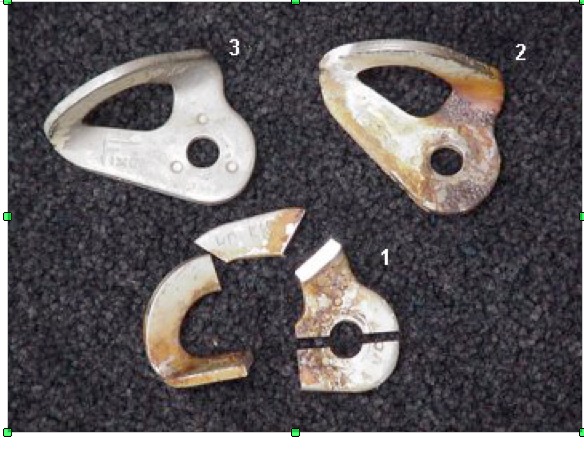
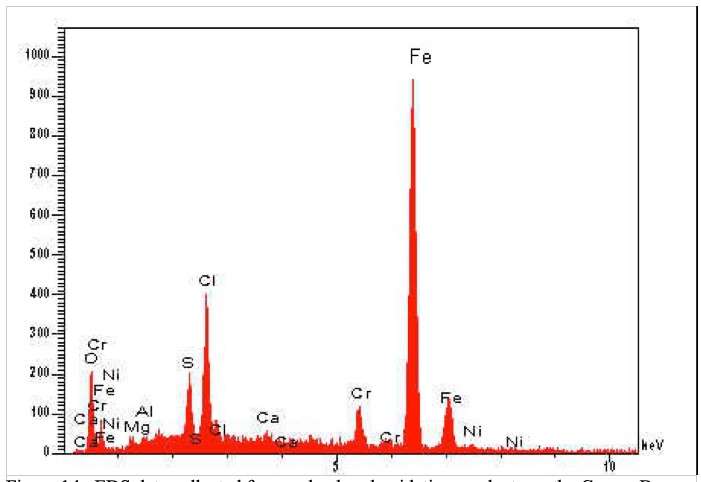
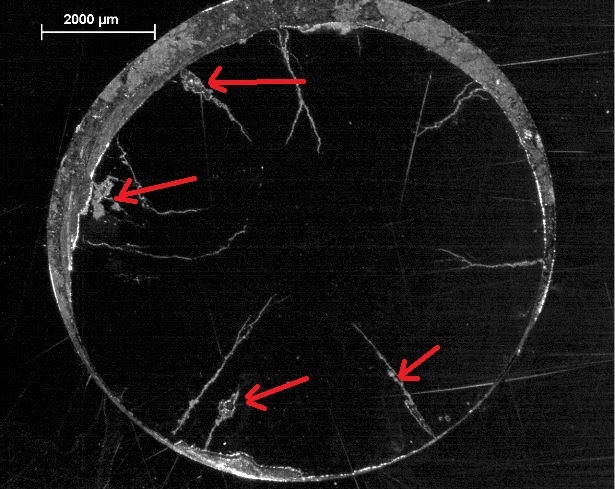
dave custerwrote: Here's what I know. This analysis was done in 2004 when not much was known about cracking bolts. The hanger in question was identified as 304 using "ICP/Spark". I've included a photo of where the hanger was cut, but I don't know for certain which surface we're looking at in the earlier photo. And the hanger, still #1, had significant surface pitting as I mentioned earlier. The large dark "pit" is the dimple stamped into the metal. You can click to enlarge photos. And the reddish oxidation product was analysed via EDS Hopefully that helps. |
|
|
Very cool. Composition by ICP would be solid, so 304 it is. |
|
|
David Reevewrote: Okay, but isn't that a contradiction? The EDS was done on the dark material on the surface, and SRBs live in crevices... ?? Leaching? |
|
|
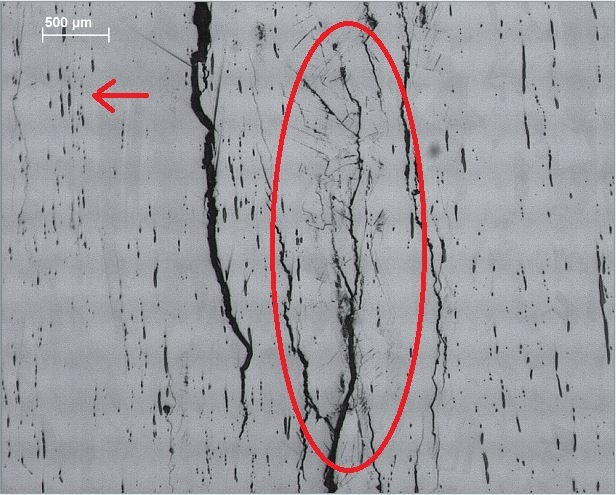
I had a brain-fart which made me think back... to 2014 and this analysis. The assumption was that the "sulphur stringers" were in the stainless steel due to poor quality control during manufacture, or that it was 303 (no ICP done) which intentionally includes S. But now it seems entirely possible that SRBs are involved. Dave, can you comment? "I sent about 10 of the broken bolts off to UCT for a decent investigation and after some cutting, polishing, etching and looking they concluded that the bolts in question were a poor quality stainless steel with sulphur stringers. Here you can see some pics showing the scary spiderweb cracks in the bolt:" http://www.climbing.co.za/forum/viewtopic.php?f=12&t=11317&p=56987&hilit=SCC#p56987 |
|
|
Wow! I haven't seen those pics before. Classic SRB attack. Firstly the stringers. These are almost certainly delta-ferrite residuals remaining following solution annealing. This is pretty much what I see in both 304 and 316 expansion bolts. The presence of a few percent of delta-ferrite is considered desirable to prevent hot-shortness of the alloy. Delta-ferrite is definitely a conduit for hydrogen diffusion, but thus far in my studies, I've found no reason to believe it plays a role in SRB attack. If you look at the stringers I've marked with the arrow, you can see that they dissolve into the austenite as part of the process of solution annealing, and often all that is left is a line of spheroidal particles. I'll stand corrected if they have analysed these residuals as sulphide, but I reckon they are just guessing. I was a bit taken aback myself when I first started looking at expansion bolts, but have since done enough reading on the subject to confirm it is pretty typical to see this level of ferrite. The region circled excites me more. I will be publishing plenty of identical pictures to this when I finally get my head around what it is I'm seeing. The etching used in preparation not only highlights what one assumes must be stress cracks, but also highlights the arrays of slip bands within the granules bordering the crack. These are likely to be zones of martensite transformation but that's a guess, that is yet to be verified. This is a big subject and only part way through looking at the samples I have from Cabo da Roca. Whatever, the explanation, we can clearly see the influence of the SRB penetrates deep into the metal. The second picture shows what I now recognize as a sign of SRB attack. That is we see areas within the crack system where the bacteria have removed substantial volumes of metal. Thus we are not dealing with a simple stress crack propagation mechanism. Abundant sulphide corrosion products, most likely mackinawite, is visible on the thread flank at the top of the image. My biggest question mark lies with, wherever does the sulphur come from? The African Plate extends way down toward the Antarctica before you encounter any tectonic volcanism. However, a bit of googling shows that a 15,000 ton sulphur stockpile in the region caught fire some 9 years previous to these bolts breaking. I wonder? |
|
|
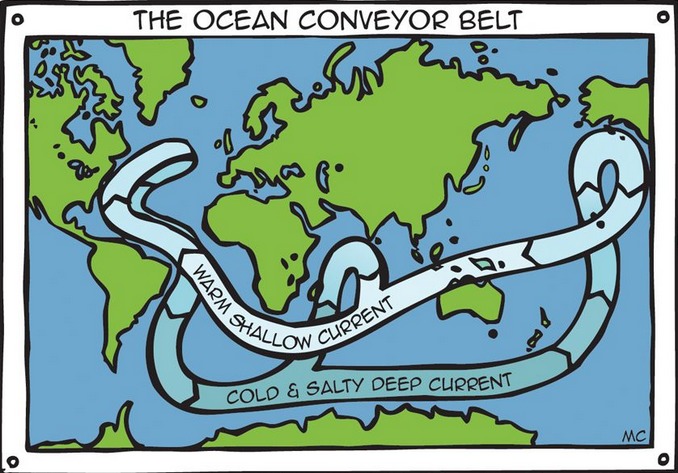
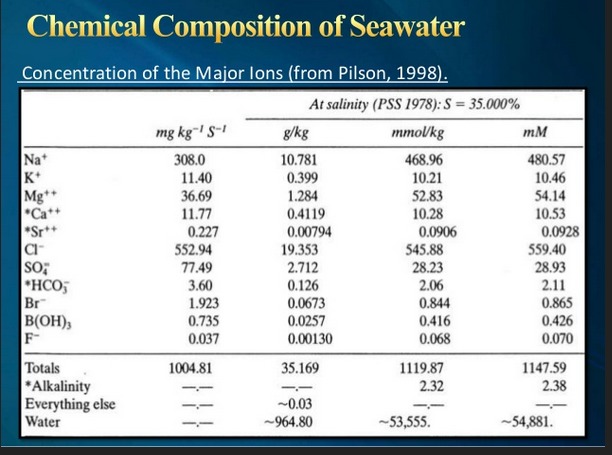
David Reevewrote: Maybe you've already provided the answer. If deep volcanic vents are dumping sulfur into the water in the Caribbean, (and elsewhere) it could be transported around the world by the Atlantic Conveyor belt, locally known as The Gulf Stream. After so much time passing and mixing, it seems seawater has high concentrations of SO4 pretty much everywhere. It looks like SO4 is the third-most common ion in the ocean after Na and Cl. So the sulpher doesn't need to have a land-based source. A reliable and large source of Sulfates has created a specialized bacteria to eat them. "Sulfate-reducing microorganisms can be traced back to 3.5 billion years ago and are considered to be among the oldest forms of microbes, having contributed to the sulfur cycle soon after life emerged on Earth.[3]" -- Wikipedia So I don't think you need a land-based source, the ocean provides all (as usual). |
|
|
David Reevewrote: Probably just the local vinyards dusting against mildew every week or so, ground-based dusting isn't very accurate when it comes to wind drift and helicopter dusting even less so. I'd look at what's being grown near the crag since hundreds of kg/hectare are being blasted over grapes, citrus crops, avacados, bananas and plenty of other plants which suffer from mildew. There's a reason someone had 15,000 tons of sulphur lying around. |
|
|
The heck with tons of the stuff; think Teragrams... https://www.nature.com/articles/s41598-018-37304-0 |
|
|
John Byrnes: It is true that all known occurrences of SRB attack are within a very short distance from the sea, and it is true that sea water carries a lot of sulphate. The level is pretty much set by the solubility of calcium sulphate. Typically, seawater is super-saturated with calcium sulphate, and thus there are recorded events where huge amounts of calcium sulphate drop out of solution to accumulate on the seabed. I puzzle over the fact that we don't see severe attack by SRB at all sea cliffs. It seems there needs to be a lot of sulphate, many times that available in sea water, to drive this process. Technical Wall in Hong Kong is the only exception to this rule we have to date. We can't locate significant sulphate levels, and I would be surprised to find any given the plate tectonics. However, it definitely has a history of chewing 304. Some of the failures can be attributed to stuff like IGC of welds, and Fixe's over-hard material problem with chain anchors. However, Tomas Prosek examined one FH that looks like SCC. It may be that this truly is SCC rather than SSC, but without a positive identification of sulphide, we don't really know one way or the other. |
|
|
Jim Titt: Good point about sulphur dusting of vineyards. I'd forgotten they did that.It could certainly cause local problems. I have an article filed away somewhere that discusses the problem of SRB for golf course management when sulphur is put down on poorly drained soil. Application of molybdate can mitigate the problem. This is part of a bigger story I'm working one, Molybdenum ions are strongly inhibitory to the sulphate reduction enzyme system of SRB. This raises the question as to whether the resistance of 316 is due to the lower diffusion rate of hydrogen by virtue of its nickel content, or the inhibition of SRB growth due to its molybdenum content. Or both... or neither :-) |
|
|
Dave Custer: For sure there is a lot of sulphur as S02 circulating in the atmosphere, but the land surface is an even bigger number, so the amount deposited is very small. Nothing like as high as the sulphate level of the sea, and even that doesn't seem to be a problem. |

 Continue with onX Maps
Continue with onX Maps Sign in with Facebook
Sign in with Facebook