|
|
John Byrnes
·
Nov 2, 2020
·
Fort Collins, CO
· Joined Dec 2007
· Points: 392
Jim Tittwrote: That she got it wrong? There's good reason why the UIAA and in Dave's article it says " despite repeated calls by UIAA Safe Com and by myself, an example of failed 316 hardware is yet to materialise". I'm reliably informed that what she examined was not the same material that she analysed. Same ole, same ole Jim. Still trying to discredit anyone who says 316 cracks in seaside placements eh? I don't know who "Dave" is, don't care. Don't know who he or the UIAA Safe Com called, or when or why. What I DO know is that your business is selling stainless steel bolts and that your motives and biases are clearly suspect. And, as has been established over and over (see above), manufacturers of stainless bolts don't always deliver the grade of steel they advertise or they conceal the grade. Or their suppliers say one thing and deliver another. You, yourself have described where a sample has been stamped "316" and was found to be 304. So you really don't know what you're getting when you buy stainless bolts and, if they corrode and fail, you are never sure why. Because of all this bullshit surrounding stainless steel, and the fact that every type of stainless bolt that has been tried in a seaside environment has failed (eg. Thailand), anyone who places stainless in seaside cliff is a fool or just evil. Titanium bolts are the proven solution to the problem.
|
|
|
Jim Titt
·
Nov 2, 2020
·
Germany
· Joined Nov 2009
· Points: 490
John Byrneswrote: Same ole, same ole Jim. Still trying to discredit anyone who says 316 cracks in seaside placements eh? I don't know who "Dave" is, don't care. Don't know who he or the UIAA Safe Com called, or when or why. What I DO know is that your business is selling stainless steel bolts and that your motives and biases are clearly suspect. And, as has been established over and over (see above), manufacturers of stainless bolts don't always deliver the grade of steel they advertise or they conceal the grade. Or their suppliers say one thing and deliver another. You, yourself have described where a sample has been stamped "316" and was found to be 304. So you really don't know what you're getting when you buy stainless bolts and, if they corrode and fail, you are never sure why. Because of all this bullshit surrounding stainless steel, and the fact that every type of stainless bolt that has been tried in a seaside environment has failed (eg. Thailand), anyone who places stainless in seaside cliff is a fool or just evil. Titanium bolts are the proven solution to the problem. What's up, have I upset you by telling you the scientific justification you put forward has been "questioned"? It was Tomas Prosek's technician that discovered her mistake so blame them for your house of cards collapsing, not me.
|
|
|
John Byrnes
·
Nov 3, 2020
·
Fort Collins, CO
· Joined Dec 2007
· Points: 392
Jim Tittwrote: What's up, have I upset you by telling you the scientific justification you put forward has been "questioned"? It was Tomas Prosek's technician that discovered her mistake so blame them for your house of cards collapsing, not me. Sorry Jim, I don't have a "house of cards". My house is built with Titanium which has never been accused of corrosion in a seaside environment by anyone. It is your house, built with stainless, that has been under attack for the last 25 years due to many, many bolt failures in seaside cliffs all over the world. Whether the failures are from SCC, SSC or some other corrosion mechanism, is academic. No one trusts it anymore, and for a very good reason. If you want to convince people that your products are safe for seaside use, PROVE IT. That is, if you can convince anyone to put stainless in a cliff in Thailand and then trust it for more than a year or two.
|
|
|
Jim Titt
·
Nov 3, 2020
·
Germany
· Joined Nov 2009
· Points: 490
John Byrneswrote: Sorry Jim, I don't have a "house of cards". My house is built with Titanium which has never been accused of corrosion in a seaside environment by anyone. It is your house, built with stainless, that has been under attack for the last 25 years due to many, many bolt failures in seaside cliffs all over the world. Whether the failures are from SCC, SSC or some other corrosion mechanism, is academic. No one trusts it anymore, and for a very good reason. If you want to convince people that your products are safe for seaside use, PROVE IT. That is, if you can convince anyone to put stainless in a cliff in Thailand and then trust it for more than a year or two. It was your idea to enter a discussion about corrosion mechanisms in 304 stainless to prosletise your titanium beliefs then claim SCC was PROVEN by quoting a paper which is reportedly utterly flawed. Then repeat your usual personal attacks, rubbishing experts who have a different opinion to yourself and resorting to wild conspiracy theories. I have as much interest in what happens in Thailand as their king, he lives 50 miles down the road from me..
|
|
|
David Reeve
·
Nov 3, 2020
·
Unknown Hometown
· Joined Jul 2020
· Points: 0
Hey look what happens when you turn your back for a few days
I'll work through a see if there are any answers I can provide. Meanwhile take a look at this pic I snapped last week. This is a from a SSC cracked bolt from Cabo da Roca. I'm pretty sure that the orange stuff is a bacterial biofilm distributed through stainless steel sponge. You can see hints of individual cells that look about the right size for Desulfovibrio vulgaris.
|
|
|
David Reeve
·
Nov 3, 2020
·
Unknown Hometown
· Joined Jul 2020
· Points: 0
Brian in SLCwrote:Interesting and nicely done report! Photo's are amazing. Those little sulpher eatin' bugs are crazy. Curious if there's any other known sea cliff locations that have sulphate and climbing with fixed anchors? To date I've analysed close to 100 samples from something like 14 different locations in the world. I've looked at a whole range of rock types and have looked at both benign and corrosive crags. The raw data were publicly viewable but I have since been taken down. I'll be republishing it on my cragchemistry.com website just as soon as I get the time.
But yeah... there are other sulphate cliffs in the world. In every case there is a correlation with the presence of undersea volcanism. Continental subduction zones are the main players, but we also see problems at points of mid-ocean volcanism, eg Canaries, Azores and Hawaii.
|
|
|
David Reeve
·
Nov 3, 2020
·
Unknown Hometown
· Joined Jul 2020
· Points: 0
Mark Frumkinwrote:What it's made out of. Nothing is impervious. 316 is what I used to hold the keel on my boat because it's more resistant to scc, ssc than 304. It's complicated but 316 may be ok because it has just sufficient nickel to stabilize the austenite during cold working. This matters because hydrogen diffusion rate is the primary enabler of severe SRB attack. It's kinda a mega read, but my argument for this theory is here ...
https://cragchemistry.com/2020/10/05/austenite-stability-are-we-missing-something-here/
|
|
|
David Reeve
·
Nov 4, 2020
·
Unknown Hometown
· Joined Jul 2020
· Points: 0
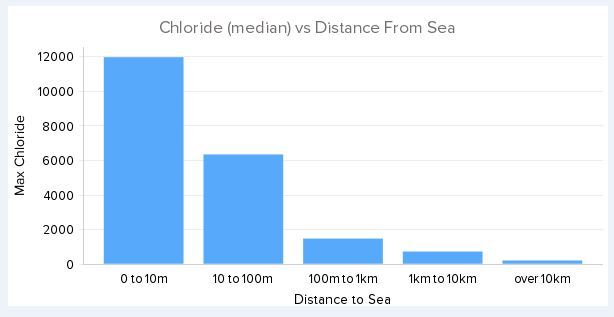
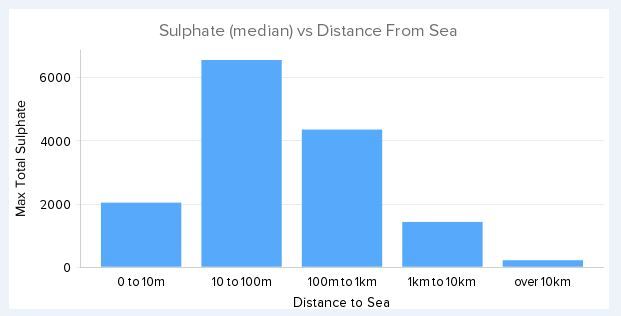
John Byrnes said - "So I've been following your research and there's a lot that's above my pay grade. However, let's look at it "backwards" for a moment. Let's say your hypothesis is correct and SRB's are causing SSC in seaside bolts. Can you then say why we don't see SSC at inland crags? It would seem reasonable to me that SRB's would be just about everywhere on the earth." Yep SRB are lurking everywhere. But there are three environmental conditions that must be met for them to flourish a) a low oxygen environment b) a source of reducing potential - steel. If an organic source is present they will leave the steel and take that instead and c) a source of oxidising potential - sulphate. The reason we don't find corrosive activity more than a few metres from the sea is because that is where the sulphate is. With few exceptions you won't find abundant sulphate inland except where volocanism is active. You also won't find it at a sea cliff either unless there is sub-sea volcanism at work. Every corrosive crag bar one that I have looked at have high levels of sulphate seeping from the ground water or coating the rocks. The level of sulphate fall off very rapidly, often within a few hundred metres from the coast.
|
|
|
David Reeve
·
Nov 4, 2020
·
Unknown Hometown
· Joined Jul 2020
· Points: 0
Re 316 SS
There are two theoretical reasons to believe 316 will be ok. By 316 I mean a material that meets the 316 elemental specification, that has been adequately soak annealed before working to reduce delta-ferrite to less than say 3%, and that has not be worked in Santa's Arctic Workshop.
a) Nickel content in excess of 10% will suppress alpha-martensite formation when the material is worked at room temperature. The formed product will be more resistant to hydrogen attack mediated by SRB. It's complicated, but I try to make it as simple as possible here https://cragchemistry.com/2020/10/05/austenite-stability-are-we-missing-something-here/
b) Significant molybdenum content raises the possibility that growth of SRB will be suppressed due to the inhibition of the enzymes involved in the sulphate reduction by molybdenum. The biochemistry of this phenomenon is well documented. I'm not convinced that the precipitation of molybdenum disulphide won't suppress such an effect. It's on the to-do list.
Meanwhile in the real world we are still looking for that failed 316 bolt. In Taiwan, the guys looking after Long Dong have a very thorough and ongoing test regime. To date they have tested many hundreds of 316 bolts that were installed at various periods from 5 to 15 years ago. They are yet to record a failure, yet this is a serious sulphur crag that absolutely eats 304.
|
|
|
John Byrnes
·
Nov 5, 2020
·
Fort Collins, CO
· Joined Dec 2007
· Points: 392
Jim Tittwrote: It was your idea to enter a discussion about corrosion mechanisms in 304 stainless to prosletise your titanium beliefs then claim SCC was PROVEN by quoting a paper which is reportedly utterly flawed. Then repeat your usual personal attacks, rubbishing experts who have a different opinion to yourself and resorting to wild conspiracy theories. I have as much interest in what happens in Thailand as their king, he lives 50 miles down the road from me.. Oh, so you can't prove it and try to change the subject. Same ole, same ole Jim.
|
|
|
John Byrnes
·
Nov 5, 2020
·
Fort Collins, CO
· Joined Dec 2007
· Points: 392
David Reevewrote:John Byrnes said - "So I've been following your research and there's a lot that's above my pay grade. However, let's look at it "backwards" for a moment. Let's say your hypothesis is correct and SRB's are causing SSC in seaside bolts. Can you then say why we don't see SSC at inland crags? It would seem reasonable to me that SRB's would be just about everywhere on the earth." Yep SRB are lurking everywhere. But there are three environmental conditions that must be met for them to flourish a) a low oxygen environment b) a source of reducing potential - steel. If an organic source is present they will leave the steel and take that instead and c) a source of oxidising potential - sulphate. The reason we don't find corrosive activity more than a few metres from the sea is because that is where the sulphate is. With few exceptions you won't find abundant sulphate inland except where volocanism is active. You also won't find it at a sea cliff either unless there is sub-sea volcanism at work. Can you clarify some things here? So SRB are anaerobic critters. However, I have experience with many cracked hangers which are exposed to air on the front side and in many cases the back-side as well. Even though the backside is a crevice and traps water, it dries out rather quickly in the sun. So is it likely that SSC is cracking them? Or SCC? I think you mean "a few hundred metres" because you say that below, right? And if I read this right, you're saying the sulphate comes from the sea-water, not the rock/land. But then in the last sentence you say you won't find it at a sea cliff which is a contradiction, isn't it? And lastly, what is the role of water/humidity in SSC? For SCC, a chemical process, low humidity is required for it to progress, but most bacteria need a wet environment to be active. Every corrosive crag bar one that I have looked at have high levels of sulphate seeping from the ground water or coating the rocks. The level of sulphate fall off very rapidly, often within a few hundred metres from the coast.
|
|
|
simplyput .
·
Nov 5, 2020
·
Unknown Hometown
· Joined Nov 2013
· Points: 60
John Byrneswrote: Oh, so you can't prove it and try to change the subject. Same ole, same ole Jim. You probably don't give a shit, but as an observer with no dog in the fight it seems to me that you come off as the one refusing to accept outside evidence that counters your beliefs or question the validity of a reportedly flawed study when the results conform to your preconceived notion. On top of that, the personalized rebukes toward Jim do not make you come off as someone interested in the scientific process, but rather as someone more interested in being right. Carry on, gentlemen...
|
|
|
Jim Titt
·
Nov 5, 2020
·
Germany
· Joined Nov 2009
· Points: 490
John Byrneswrote: Oh, so you can't prove it and try to change the subject. Same ole, same ole Jim. Can't prove what?
|
|
|
David Reeve
·
Nov 5, 2020
·
Unknown Hometown
· Joined Jul 2020
· Points: 0
John Byrneswrote: Can you clarify some things here? So SRB are anaerobic critters. However, I have experience with many cracked hangers which are exposed to air on the front side and in many cases the back-side as well. Even though the backside is a crevice and traps water, it dries out rather quickly in the sun. So is it likely that SSC is cracking them? Or SCC? I think you mean "a few hundred metres" because you say that below, right? And if I read this right, you're saying the sulphate comes from the sea-water, not the rock/land. But then in the last sentence you say you won't find it at a sea cliff which is a contradiction, isn't it? And lastly, what is the role of water/humidity in SSC? For SCC, a chemical process, low humidity is required for it to progress, but most bacteria need a wet environment to be active. Yes SRB are obligate anaerobes, and yes they need water. So under the dry environment of the Mediterranean and the Portuguese coast, it is rare to see corrosion on the exterior surface of a bolt. There is a tendency toward the very opposite because chloride pitting is suppressed in the presence of sulphate. However, at a point below the hanger, a few mm into the rock, the conditions for low oxygen, moisture and high sulphate are met, and SRB will flourish. When we do see exterior signs of corrosion, it has always broken through from underneath.
However, under the high humidity conditions prevailing at Railay, something different happens. It may be the bacteria is different, or it may be the same bacteria responding to a different environment, but what we see is the development of SRB colonies on the external surfaces of the metal in addition to the usual development at glue-lines or beneath nuts, washers etc. The development of external colonies is a common occurrence on the outer radius of sharp bends of ring bolts. My guess is that micro-cracks are providing a low oxygen space for the initiation of bacterial colonies. I say colonies, because, it is well documented that for conditions of high-humidity or those under-water, the bacteria form an oxygen-excluding iron oxide/sulphide 'igloo' over the corrosion pit that forms.
Have you read my post on Tonsai anchor rings? The evidence points to SRB involvement, although the sulphate supply looks to be patchy.
https://cragchemistry.com/2020/04/09/a-certain-ring-of-truth/
With respect sulphate availability, yes the source is the sea and the availability falls-off very rapidly with distance from the sea.
The attenuation of sulphate and chloride by vegetation, and by gravitational fall-out, suggests the carrier is the coarse marine aerosol in the particle size range of say 20um to 60um. I have a data set of close to 100 wall-wash samples from maybe 14 different crags across the world - some inland come coastal. The attenuation curve of sulphate does not match that of chloride and thus we know that the source of the sulphate is not the sulphate dissolved in the seawater, but must be biogenically formed from a different marine sulphur source. My current theory is that the source is elemental sulphur being carried by the water-insoluble, living scum that covers the surface of the oceans. When elemental sulphur is deposited on the thin soils of ledges and the clifftop proper, sulphur oxidising bacteria will convert it to sulphuric acid, and thus we have sulphate running down the rock surfaces and entering the immediate ground water.
Why don't we have this problem at all sea-cliffs? Simply because the release of elemental sulphur into the sea is a relatively rare phenomenon. All sulphate crags are associated with regional sub-sea volcanism. No regional volcanism, no problems.
![]() ![]()
|
|
|
John Byrnes
·
Nov 6, 2020
·
Fort Collins, CO
· Joined Dec 2007
· Points: 392
simplyput .wrote: You probably don't give a shit, but as an observer with no dog in the fight it seems to me that you come off as the one refusing to accept outside evidence that counters your beliefs or question the validity of a reportedly flawed study when the results conform to your preconceived notion. My preconceived notion? Angel's peer-reviewed article was published in a prestigious professional journal in 2008. Her first reference was my article published in 2001. Her bona fides: "Angele Sjong, PhD Senior Chemical Reliability Consultant Angele is a senior Chemical Reliability Engineer with Ops A La Carte and president of Sjong Consulting LLC. Sjong Consulting partners with Ops A La Carte in the areas of materials engineering, polymers and chemistry. Angele holds Ph.D. degree in Inorganic Chemistry from Yale University and M.S. in metallurgical engineering from the Colorado School of Mines. She has been a technical consultant on issues related to product liability, patent infringement and failure analysis. She has provided technical consulting services to companies on pressure vessels and piping; consumer electronics;recreational equipment; consumer products; and automotive equipment. She has been an expert witness for litigation cases involving ferrous metallurgy, corrosion and glass. Angele has also been active with ASM, and taught a polymers failure analysis class to medical device engineers." She has 5 pages of patent applications, ~20 patents per page, going back to 2009: https://patents.justia.com/inventor/angele-sjong ------------------------- Besides her findings, there is a large body of evidence gathered over 20 years, much of it by the UIAA, that shows 304 and 316 cracks in both the laboratory and the cliff. "Recently the UIAA website has published an extensive document in both English and French that highlights the results and the conclusions the researchers have come to after years of work. Unfortunately, according to the text linked below, it seems as if only titanium (and therefore not even High Corrosion Resistant (HCR) steels) are suitable for crags located in places classified as being high risk." https://www.planetmountain.com/en/news/climbing/uiaa-recommends-titanium-bolts-only-for-marine-crags.html ------------------------- So when Jim, who does have a dog in the fight, insinuates without proof that Angele and the UIAA are wrong, should I just believe him? Or should I ask him to produce his evidence? On top of that, the personalized rebukes toward Jim do not make you come off as someone interested in the scientific process, but rather as someone more interested in being right.
Really? Maybe after so many years, I'm just tired of Jim trying to discredit anyone who says stainless should not be used for sea cliffs. I respect his knowledge and his willingness to share it here but I'm really tired of his continued attacks.
|
|
|
John Byrnes
·
Nov 6, 2020
·
Fort Collins, CO
· Joined Dec 2007
· Points: 392
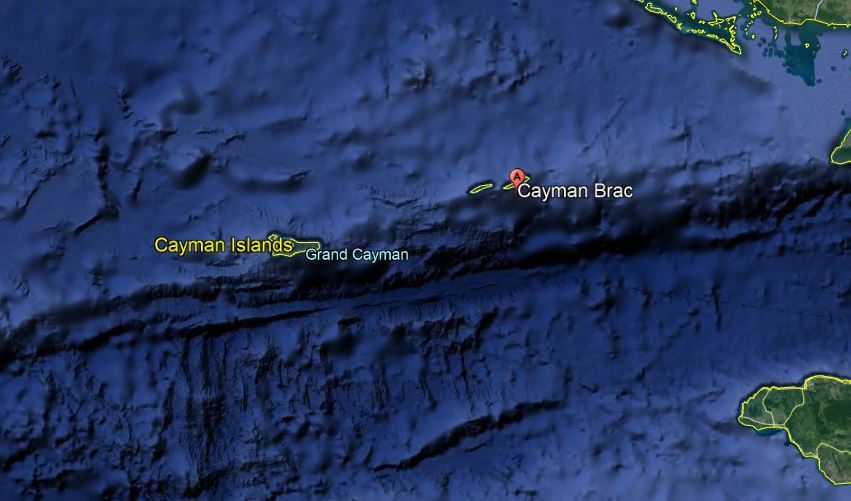
David Reevewrote: Yes SRB are obligate anaerobes, and yes they need water. So under the dry environment of the Mediterranean and the Portuguese coast, it is rare to see corrosion on the exterior surface of a bolt. There is a tendency toward the very opposite because chloride pitting is suppressed in the presence of sulphate. However, at a point below the hanger, a few mm into the rock, the conditions for low oxygen, moisture and high sulphate are met, and SRB will flourish. When we do see exterior signs of corrosion, it has always broken through from underneath. Interesting. In Cayman Brac I've seen lots of corrosion on outside surfaces (photo of cracked hanger), as well as the corrosion at the line between oxygen/no-oxygen just inside the rock that you mention. Is there a name for that line? The first thing that comes to my mind is that it might be possible for SSC and SCC to be "collaborating" in the cracking of the bolts and hangers. In the SEM photo below, the cracks were identified as resulting from SCC. Do SSC cracks look similar? ![]()
However, under the high humidity conditions prevailing at Railay, something different happens. It may be the bacteria is different, or it may be the same bacteria responding to a different environment, but what we see is the development of SRB colonies on the external surfaces of the metal in addition to the usual development at glue-lines or beneath nuts, washers etc. The development of external colonies is a common occurrence on the outer radius of sharp bends of ring bolts. My guess is that micro-cracks are providing a low oxygen space for the initiation of bacterial colonies. I say colonies, because, it is well documented that for conditions of high-humidity or those under-water, the bacteria form an oxygen-excluding iron oxide/sulphide 'igloo' over the corrosion pit that forms.
Have you read my post on Tonsai anchor rings? The evidence points to SRB involvement, although the sulphate supply looks to be patchy.
https://cragchemistry.com/2020/04/09/a-certain-ring-of-truth/
Yes. So one hypothesis is that SCC is causing micro-cracks in the surface during low-humidity periods, then SSC moves in and finishes the job during the usual high-humidity. Thoughts on that idea? Could explain why the metal cracks so rapidly in some cases. With respect sulphate availability, yes the source is the sea and the availability falls-off very rapidly with distance from the sea.
The attenuation of sulphate and chloride by vegetation, and by gravitational fall-out, suggests the carrier is the coarse marine aerosol in the particle size range of say 20um to 60um. I have a data set of close to 100 wall-wash samples from maybe 14 different crags across the world - some inland come coastal. The attenuation curve of sulphate does not match that of chloride and thus we know that the source of the sulphate is not the sulphate dissolved in the seawater, but must be biogenically formed from a different marine sulphur source. My current theory is that the source is elemental sulphur being carried by the water-insoluble, living scum that covers the surface of the oceans. When elemental sulphur is deposited on the thin soils of ledges and the clifftop proper, sulphur oxidising bacteria will convert it to sulphuric acid, and thus we have sulphate running down the rock surfaces and entering the immediate ground water.
Why don't we have this problem at all sea-cliffs? Simply because the release of elemental sulphur into the sea is a relatively rare phenomenon. All sulphate crags are associated with regional sub-sea volcanism. No regional volcanism, no problems.
![]() ![]()
I gotta think more on this aspect. Cayman Brac is a tall basalt tower (~7,200 metres high) with a limestone cap. There are also layers in certain areas of "Caymanite" which is thought to be formed by deposited ash eons ago. So there was volcanism, but not recently. It is also subjected to frequent hurricanes, unlike the Med, which blow sea water spray and aerosols over the entire island (it's only 1.5km wide). It has fresh water wells and "brackish" wells which have high concentrations of Hydrogen Sulphide, H2S. Hmmm...
|
|
|
David Reeve
·
Nov 6, 2020
·
Unknown Hometown
· Joined Jul 2020
· Points: 0
John Byrnes: "Interesting. In Cayman Brac I've seen lots of corrosion on outside surfaces (photo of cracked hanger), as well as the corrosion at the line between oxygen/no-oxygen just inside the rock that you mention. Is there a name for that line? "
Those cracks certainly could be SSC. I don't believe there is any way we can tell just by looking. However, the black corrosion deposits would have me reaching for my sulphide spot test. I don't know of any particular name being given to the boundary between high and low oxygen except to note that a such sharp boundary is a well documented feature of biofilms. Sulphur is often closely conserved with sulphate reducers in the inside and sulphide oxidisers on the outside.In such structures sulphur cycles from inside to outside and back with iron being removed with every cycle. Of course this interface tends to be marked by the "black ring of death" as you so aptly named it.
John Byrnes: "The first thing that comes to my mind is that it might be possible for SSC and SCC to be "collaborating" in the cracking of the bolts and hangers. In the SEM photo below, the cracks were identified as resulting from SCC. Do SSC cracks look similar?"
It is unlikely that chloride doesn't play a role. If it is present, which it is in substantial concentrations, then it is part of the electro-chemical mix. However, SCC is considered an anodic mechanism, whilst SSC is cathodic. As they are proposed, the mechanisms of crack propagation are dissimilar, although some authors blur the distinction by assigning a role to atomic hydrogen in both. Crevice corrosion, however, is a process resulting from a depletion of low oxygen. I could conceive of that as an entry point for SRB.
As for the photos of the cracks in the cross-section, I reckon the identification of SCC is wrong. The crack propagation direction should give pause for thought. This pattern is typical of stress cracks running into the metal from an internal SRB colony. SSC cannot nucleate from within the bulk metal, and thus will show clear crack development that propagates from the outer surface orthogonal to the primary stress vector.
![]()
|
|
|
David Reeve
·
Nov 6, 2020
·
Unknown Hometown
· Joined Jul 2020
· Points: 0
John Byrne: "I gotta think more on this aspect. Cayman Brac is a tall basalt tower (~7,200 metres high) with a limestone cap. There are also layers in certain areas of "Caymanite" which is thought to be formed by deposited ash eons ago. So there was volcanism, but not recently. It is also subjected to frequent hurricanes, unlike the Med, which blow sea water spray and aerosols over the entire island (it's only 1.5km wide). It has fresh water wells and "brackish" wells which have high concentrations of Hydrogen Sulphide, H2S. Hmmm... "
I'm yet to find a case where plate tectonics were not the most plausible root cause of crag sulphate. In the case of Cayman Brac, you have an active trench opening right offshore. It has "smokers" pumping sulphur into the deep ocean. Sulphur eventually floats to the surface, and becomes part of the insoluble bio-scum that floats on the ocean surface. Wave break and strong winds carry this as coarse aerosol to cliff tops and a short distance inland. Sulphur is oxidised to sulphate and, yeah, you are going to get sulphide build up in stagnant waters.
From Wikipedia - Cayman Trough
In 2010 a UK team from the National Oceanography Centre in Southampton (NOCS), equipped with an autonomously controlled robot submarine, began mapping the full extent of the trench and discovered black smokers on the ocean floor at a depth of 5 km (3.1 mi), the deepest yet found.[5][6][7] In January 2012, the researchers announced that water exits the vents at a temperature possibly exceeding 450 °C (842 °F), making them among the hottest known undersea vents. They also announced the discovery of new species, including an eyeless shrimp with a light-sensing organ on its back.[8]
![]()
|
|
|
dave custer
·
Nov 8, 2020
·
Unknown Hometown
· Joined Nov 2010
· Points: 3,078
...
As for the photos of the cracks in the cross-section, I reckon the identification of SCC is wrong. The crack propagation direction should give pause for thought. This pattern is typical of stress cracks running into the metal from an internal SRB colony. SSC cannot nucleate from within the bulk metal, and thus will show clear crack development that propagates from the outer surface orthogonal to the primary stress vector.
This pattern looks similar to the fractures I expect in sheer loading. I can't tell which cross section I'm looking at and whether there is some obvious reason for there to be a sheer stress that would cause the cracks to propagate on the diagonal. I find these cracks to be rewarding when they are in rock and not the anchors, e.g. cdn2.apstatic.com/photos/cl…
|
|
|
David Reeve
·
Nov 8, 2020
·
Unknown Hometown
· Joined Jul 2020
· Points: 0
Yeah, I was wondering about the diagonal orientation as well. I think it is a section of a FH. There are the usual stringers of delta ferrite showing the rolling direction of the plate stock, and the upward deflection of these on the far right edge looks very much like this is a pressed edge, punched from lower to upper. I reckon it has to be residual stress from manufacture, but I'd need to see the whole thing to make sense of it.
|

 Continue with onX Maps
Continue with onX Maps Sign in with Facebook
Sign in with Facebook